CE 457/527
Colloid and surface phenomena aspects of Chocolate
DATE OF SUBMISSION: 04-09-2002
GROUP MEMBER: TECK YU SIA
CHIN KOK OOI
SHUSHAN MUNSHI
ANSHU VERMA
TABLE OF CONTENTS
I.
Introduction 3
II.
Components and
compositions 5
III.
Chocolate manufacturing 9
IV.
Chocolate flow
properties 14
V.
Surface- active
substances in chocolate manufacturing 20
VI.
Packaging and storage 26
VII.
Marketing 31
VIII.
Reference Cited 33
I. Introduction
United States itself consumes approximately 3 billion pounds of chocolate every year. Chocolates are common in almost any local convenience store and a wide variety of chocolates exist in the market. Although everyone seems to be familiar with the taste of chocolate, the knowledge of process technology used in manufacturing is not well known among common people. In this project, the engineering aspects, starting from the components and composition to packaging, will be examined.
To stay in the market and meet the consumer need, chocolate needs to fulfill three requirements. To attract the consumer, the chocolate has to have a uniform appearance both of external surface and cross-section when the chocolate is cut or broken. Chocolate should melt readily in the mouth to give a smooth “creamy” taste. Finally, the chocolate should be dry when touched and should have a degree of brittleness so that it will break easily under pressure from fingers. Only through proper processing, treatment, and packaging, this goal is attained. Chocolates that undergo this process, usually lasts one to ten years depending on the type of chocolate.
Types of chocolate include chocolate liquor, semi-sweet chocolate, milk chocolate, sweet chocolate, and white chocolate. Each of the above mentioned types have slightly different processing methods and ingredients. However, the vital ingredient found in all types of chocolate is cocoa butter. The chocolate liquor contains about 53 percent cocoa butter and is produced by grinding the center of the cocoa bean. By adding extra ingredients to this substance, different types of chocolates are produced. The sweet chocolate is similar to the dark chocolate but has more sweeteners. The semi-sweet chocolate is known as the dark chocolate and must contain at least 35 percent chocolate liquor. Adding milk, sweeteners, and flavorings to the chocolate liquor produces milk chocolates. The white chocolate contains more milk solids and flavorings than the milk chocolate and has no non-fat cocoa solids.
People generally tend to believe that chocolate is the cause of headaches, cavity, and high level of cholesterol. Through studies and research, it has been proven that those “myths” are false. People have complained of headaches or migraines after eating chocolate. However, it’s the lifestyle, genetics, medication, and the food sensitivity of the person that causes headaches. Chocolate is also not the primary source of cavity. Cavity is formed from the sugar and starch embraced through out the day. It is less likely to get cavity by brushing the teeth thoroughly or rinsing with water.
It is natural to believe that the saturated fat in the cocoa butter of the chocolate increases the blood cholesterol level. Not all types of saturated fats raise blood cholesterol level. The saturated fat found in the cocoa butter does not raise the LDL-cholesterol level and increases the HDL-cholesterol level. HDL-cholesterol is the “good” cholesterol and LDL-cholesterol is the “bad” cholesterol, which people with cholesterol problem should avoid. Research has also shown that chocolate contains antioxidants that may reduce the risk for developing heart disease and offer some anti-aging health benefits.
The study is still going on to produce a better quality chocolate by using different kinds of cocoa bean, different process method, and different chemicals for coating and preservation.
II. COMPONENTS AND COMPOSITIONS
Everyone is familiar with the taste of chocolate, and it is liked by everyone because of its pleasant flavor and texture, which is hard to bite and yet melts easily in the mouth. Since the process of making chocolate has been systematized, it is important to know what chocolate is made of, in order to understand what makes it so special. The essential ingredients of chocolate are cocoa, cocoa butter, milk and sugar.
Cocoa and cocoa butter are both obtained from the seeds of the Theobroma Cocoa, also know as ‘cocoa beans’, which grow in pods on cocoa trees in tropical countries. The pods are egg-shaped and are about eight inch long, and contain about twenty to thirty beans that are embedded in a soft white starchy pulp. The beans are slightly larger than coffee beans, and the color may be white or pale purple. The most important constituent of the cocoa bean are the fat, which makes the bulk of the bean, protein, starch, theobromine and caffeine, and a peculiar pigment known as ‘cocoa red’. [1]
|
Composition |
Per cent |
Composition |
Per cent |
|
Moisture |
6.3- 8.5 |
Cocoa Red |
2.5- 5.0 |
|
Fat |
46.9- 52.1 |
Ash |
2.9- 4.8 |
|
Albuminoids |
11.6- 21.1 |
Astringent matters |
7.2- 8.6 |
|
Cellulose |
3.3- 6.6 |
Cane sugar |
|
|
Alkaloids |
0.3- 0.5 |
Starch |
8.7- 12.6 |
Table II.a General composition of cocoa bean[2].
There are two forms of theobromine found in the bean, one is partly in combination with dextrose, glucoside and tannate, and the other one is free theobromine. Chemically, theobromine is closely related to caffeine. This close relationship could be told by their structure, where theobromine is 3,7-dimethyl-xanthine while caffeine is 1,3,7-trimethyl-xanthine.[3] Both of these compounds are alkaloids, and are related to the taste of bitterness of cocoa and chocolate. The peculiar and interesting pigment, the ‘cocoa red’, is formed during the drying of the beans by the action of enzymes on the glucosides.[4] This special pigment is important to the manufacturer, since it determines very largely the color of the finished product. After series of processing, the hulled and germed beans are called nibs. The nibs contain fifty per cent of fat, which is known as cocoa butter.
|
Triglycerids |
Mol, % |
|
|
Saturated acids -- Palmitostearins |
2 |
2 |
|
One unsaturated acid – Oleo-palmitostearin |
52 |
|
|
Oleo-distearin |
19 |
77 |
|
Oleo-dipalmitin |
6 |
|
|
Two unsaturated acids – Palmito-diolein |
9 |
21 |
|
Stearo-diolein |
12 |
|
Table II.b The composition of cocoa butter[5]
A large amount of fat is required in chocolate manufacturing because it helps chocolate melt smoothly in the mouth. Moreover, the special feature of cocoa butter that makes chocolate hard and brittle at normal room temperature but melts in the mouth is that the melting point of cocoa butter is between 32oC and 36oC, which is close to human body temperature. This special characteristic also helps to prevent uncontrolled fat crystallization that may result in giving chocolate an impaired texture and a gray surface appearance referred to as ‘fat bloom’. [6]
Milk is widely used in chocolate manufacturing to produce various types of milk chocolates and also to contribute to flavor, texture and color in the final product. The major components of milk are fat, proteins, lactose, and minerals.
|
Component |
Content (% w/w) |
|
Water |
87.3 |
|
Proteins (caseins & whey proteins) |
3.3 |
|
Fat |
3.9 |
|
Lactose |
4.6 |
|
Minerals |
0.65 |
|
Others (vitamins, organic acids, etc) |
0.32 |
Table II.c Composition of cow’s milk[7]
Besides the major components, milk also contains many different substances of high nutritional value, such as vitamins. For this reason milk, as an ingredient, in general enhances the nutritional value of chocolate. Milk protein composes of about eighty per cent caseins and twenty per cent whey proteins. “Caseins are flexible proteins, which exist predominantly as colloidal casein micelles, and they are remarkably heat stable. Whey proteins are globular proteins, which can be denatured by heat.”[8] Both of them are important and related to the characteristics of the finished product. The major components in milk fat are triglycerides.
Component |
Content (% of
total fat) |
|
Triglycerids |
96-99 |
|
Diglycerides |
0.3-1.6 |
|
Monoglycerides |
0.02-0.1 |
|
Free fatty acids |
0.1-0.4 |
|
Phospholipids |
0.2-1.0 |
|
Sterols |
0.2-0.4 |
|
Cerebosides |
0.01-0.07 |
Table II.d Typical composition of lipids in milk[9]
The minor surface-active constituents like mono- and diglycerides and phospholipids in milk can have a great impact on the physical properties of milk fat and need to be taken into account when milk is used in chocolate manufacture.[10] Lactose has a low solubility and can exit either in the amorphous state or in crystalline states. Lactose acts as a carrier of flavor and also contributes in imparting the right color to chocolate. Besides, the crystalline structures of lactose will influence the particle size distribution of the final product.
Sugar, an important food ingredient, is widely used in chocolate manufacture. Sugar is mainly made from sugar cane or sugar beet. Both of them give an absolutely identical natural substance, which is chemically known as ‘sucrose’. Sucrose is a white crystalline solid and is a combination of one unit of ‘glucose’ and one unit of ‘fructose’.[11] Normally, sugar industries supply a wide variety of sugars, such as monosaccharides. Among these, sucrose is the most important sugar that is used in the production of chocolate. However, in using sugar for chocolate manufacturing, one must be aware of the problem of ‘sugar bloom’. Sugar bloom occurs on chocolate surfaces from crystallization of sugar under poor temperature and humidity conditions.[12] Sugar bloom also leaves a ‘sandy’ or gritty taste in chocolate.
III. CHOCOLATE MANUFACTURING PROCESS
The making of chocolate involves many processes, which in turn are critical in producing this unique product, with its pleasant flavor and texture, which is hard to bite and yet melts easily in the mouth. These processes are outlined in (Fig III.a). Each process is important regarding the development of flavor and the improvement of the colloidal properties of chocolate. In this report, however much attention shall be given to the colloidal aspects and just a brief mention about the development of flavor during these processes.
1. Fermentation and Drying: Fermentation is done by heaping the beans and covering them with leaves. This removes adhering pulp and mucus, kills the germ of the bean, and also modifies the flavor and color of the bean. After fermentation the beans are sun-dried or machine-dried to about 7% moisture to give them good keeping quality. Fermentation and drying also alter the seed coat, which can now be easily removed.1
2. Roasting:
This process is critical in determining the correct flavor. The roasting
temperature and duration have a significant effect, as does the moisture
content of the bean and the surrounding air. Usually, whole beans are roasted
at temperatures of about 105-145 oC for an hour. This leads to the
development of many of the chemicals required to give the chocolate its desired
flavor, together with some unpleasant flavors, which are removed by subsequent
processes. In addition it loosens the shell around the bean, making the shell
easier to remove. 2
3. Winnowing:
The beans are passed through winnowing machines which remove seed coats and
separate germs. The hulled and germed beans are now called nibs. Broken beans are size selected on vibrating
sieves, before being subjected to rapidly rising currents of air. The light
platelet-shaped shell particles tend to rise. They are filtered off, where as
the denser round pieces of nib fall into collecting channels.2
4. Milling:
The nibs are passed through mills where they are torn apart and ground, in
order to release fat from the cells. The fat melts and the ground nibs acquire
a liquid consistency. The mill discharges a liquid, which is known as chocolate
liquor.3
5. Chocolate
liquor: The main components of chocolate liquor are 55% fat, 17%
carbohydrate, 11% protein, 6% tannin compounds, 3% ash, 2.5% organic acids, 2%
moisture, traces of caffeine, and about 1.5% theobromine, an alkaloid related
to caffeine that is responsible for the mildly stimulating properties of cocoa
and chocolate.4
6. Cocoa
manufacture: A portion of the chocolate liquor goes in the manufacture of
cocoa. The advantage of this process in chocolate manufacturing is that it
provides it with the essential cocoa butter. Milk chocolates are made from
chocolate liquor, but it needs additional fat (cocoa butter) that comes from
this process to coat the remaining milk and/or sugar particles.5
7. Chocolate
grinding: The production of chocolate liquor ensures that the cocoa powder
particles are small enough to produce chocolate. However, the sugar still
requires grinding. Sugar is grounded into very fine particles so that the
surface becomes very hot and changes from a crystalline to a glassy structure.
During this short period, the surface becomes chemically reactive and absorbs
many flavors (mainly cocoa flavor) and enhances the taste of the final product.
It ultimately reverts to crystalline structure. This is the advantage of
combined milling over separate milling.5
8. Conching: During conching, both chemical and physical changes take place. However, the physical changes that lead to the reduction in viscosity shall be given more importance in this report. Conching is a mixing process, which takes place in special heated tanks. The chocolate is kneaded to improve its viscosity. As the temperature rises, the fat becomes more liquid and starts to coat the solid particles. Viscosity is reduced by making further cocoa butter and emulsifier (usually lecithin) additions. The chocolate very rapidly becomes thinner and can be easily pumped to storage tanks.5 A lot more about these emulsifiers and how they modify properties shall come in the following chapter.
Tempering: This is a pre-crystallization
process. “Just as carbon atoms group together in different ways to produce
substances with a wide range of textures and surface properties (graphite to
diamond), cocoa butter also crystallizes in six different forms (Fig III.b).
Forms I to IV would produce a crumbly product, without any
gloss or snap. The fat would also soon migrate to surface, giving a white sheen
known as chocolate ‘bloom’. In addition, the chocolate expands initially upon
setting, as the crystals are not tightly packed. This means that, if a liquid
chocolate is in the wrong crystalline form when it is poured into a mould, it
is very difficult to get the solid product to come out again. For this reason
the chocolate must be seeded with form V crystals. Form VI does not
normally crystallize from the liquid chocolate but it is a solid-solid
transformation with time, also giving rise to chocolate bloom as it does so.
Devices, which are generating these seed crystals are known in the industry as
a “temperer.” 6
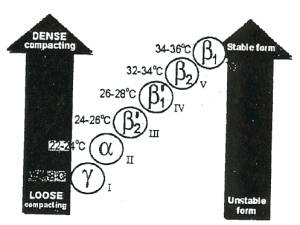
Figure III.b : Illustration of the different crystalline states of
cocoa butter. Form V is the one
required for chocolate making
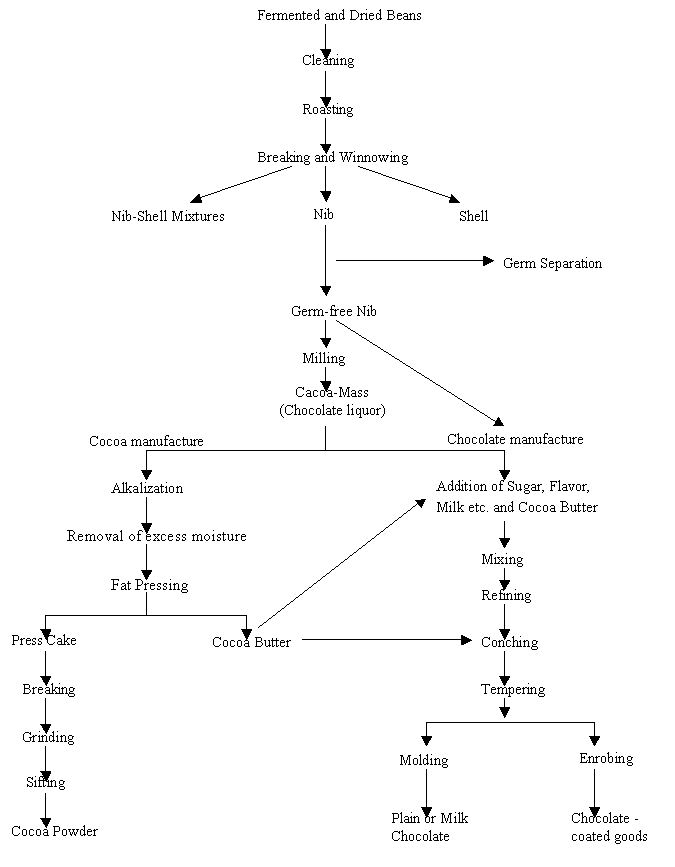
FIG III.a A flow sheet of cocoa and chocolate manufacturing
plant operations. Courtesy Food Science (Fourth edition)
IV. CHOCOLATE
FLOW PROPERTIES
1
“Molten chocolate is a suspension of particles of sugar, cocoa and/or milk solid in a continuous fat phase. Because of the presence of solid particles in the melted state, it does not behave as a true liquid but exhibits non-Newtonian flow properties.” In a plot of shear rate (D) versus shear stress (t), three different rheogram can be obtained.(see Fig IV.a).
D
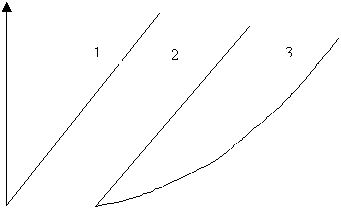
For Newtonian fluids,
t = h D
where h is the viscosity.
For Bingham fluids,
t = to + hpl D
where, to is the yield value and hpl is the plastic viscosity.
“In curves (2) and (3), if shear stress (t) is divided by the shear rate (D), without taking yield value into account, a derivative called the apparent viscosity is obtained.
Amongst the various attempts to express the curvature of the chocolate rheogram in terms of an equation, the most successful was that of Steiner (1), who, in 1958, adapted a model proposed by Casson for printing ink. Steiner’s model for chocolate was adopted by the OICC (Office International du Cacao et du Chocolat) in 1973(2) and is now accepted by most of the authors.”
The following relationship, given by Steiner (1), relates shear stress (DN) to other parameters:
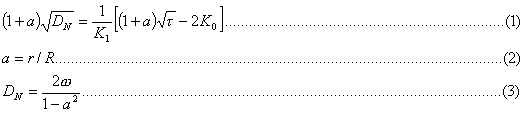
where r, R are the inner and outer radius of the cylinder respectively, w is the angular velocity, DN is the shear rate at the inner cylinder.
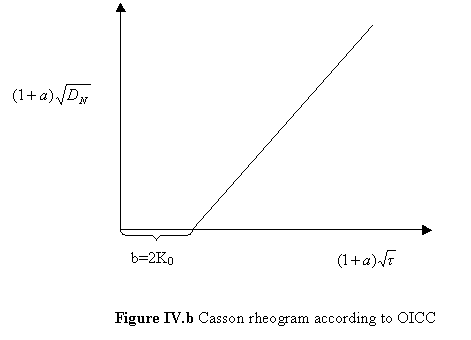
The graphical plot of equation (1) is given in Fig IV.b .
hCA = (1/slope)2 = (1/K1)2 = plastic viscosity according to Casson;
tCA = (b/2)2 =K02 = yield value according to Casson
FACTORS AFFEECTING THE FLOW PROPERTIES OF CHOCOLATE
From the literature , the Casson parameter ranges for chocolates are:
hCA = 1 to 20 Pa s
tCA = 10 to 200 Pa
Fat content, emulsifier content (e.g. lecithin), moisture content, particle size distribution, temperature, conching time, temper, thixotropy and vibration influence these values. In this section the influence of these factors will be discussed.
1. Fat content: “Dilution of a particulate suspension with the liquid which forms its continuous phase (in the case of chocolate: cocoa butter with or without milk fat) reduces its viscosity”. It means that fat acts as a lubricant for the system.Fig IV.c
2 1
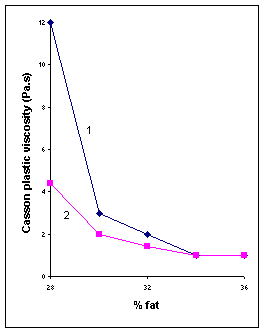
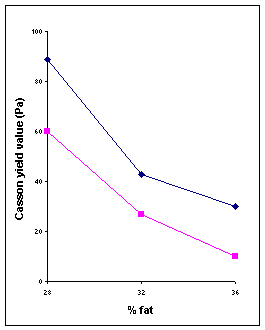
Fig
IV.c Influence of fat content on Casson parameters of two milk
chocolates with 0.25% lecithin.(1) Fine chocolate with 5.7% particles >
20mm;
(2) moderaltely coarse chocolate with 16% particles > 20mm.
2. Lecithin
and other emulsifiers: Small amount of suitable surface-active lipids can
produce an immediate reduction in viscosity. The addition of such surfactants
is very critical to the chocolate industry and hence it will be dealt in great
detail in the next section.
3. Moisture
content: There is an increase in the viscosity of chocolate with
increase in moisture content. This could be explained by the fact that
moisture tends to make the surface of sugar particles sticky and thereby
increases friction.
4. Particle
size distribution: Casson yield value increases as the particles are made
smaller. The formation of bonds or the increase in frictional contact because
of increased surface area is perhaps responsible for this behavior. The minimum
in the plastic viscosity appears due to the fact that it corresponds to maximum
packing efficiency.
2 1
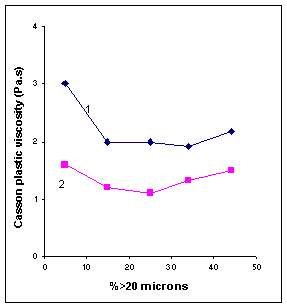
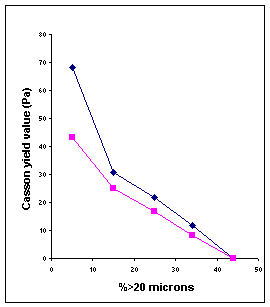
Fig
IV.d Influence of fineness on Casson parameters of two milk chocolates
with 0.25% lecithin. (1) 30% fat; (2) 32% fat
5. Temperature: As temperature increases, two phenomena are observed in milk chocolates
a.) Casson plastic viscosity (hCA) decreases.
b.) Casson yield value (tCA) may increase, especially for chocolates without lecithin.
This thickening effect can be reduced by lecithin addition, especially in the case of
dark chocolate.
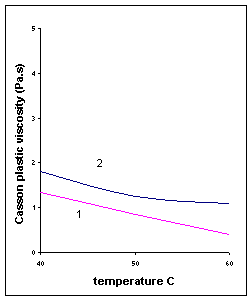
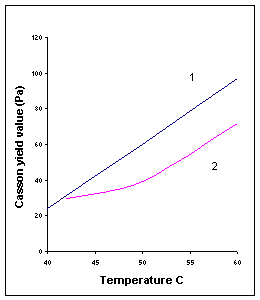
Fig
IV.e Influence of temperature on Casson parameters of two milk
chocolates. (1) 34% fat, without lecithin; (2) 30% fat, 0.15% lecithin
6. Temper: During tempering, the amount of solid particles is slightly increased and hence the viscosity. In this state that chocolate is used for molding. “However, according to Rohan and Stewart, for a well-tempered chocolate, the amount of seed crystal is relatively low and the difference in apparent viscosity before and after tempering (temperature being held constant, at about 30oC) is low”. Over-tempering leads to a significant increase in viscosity.
7. Thixotropy: the property of various substances of becoming fluid when disturbed. It is the decrease of apparent viscosity with stirring time at a given rate of shear.
8. Vibration: Vibration can influence the flow properties of chocolate during the molding and coating operation. “Bartusch showed that apparent viscosity (at a slow shear rate of 1 s-1) decreased with increasing amplitude of vibration and he postulates that the yield value had disappeared”. Vibration helps chocolate to spread in moulds. In this case the determining factor is the product of vibration amplitude and frequency.
V. SURFACE-ACTIVE
SUBSTANCES IN CHOCOLATE MANUFACTURING
Lecithin is the most commonly used surfactant in chocolate. Even a small addition of about 0.3% can produce a huge reduction in viscosity of approximately 50%. In the absence of lecithin a similar reduction in viscosity is achieved by the addition of 4% of cocoa butter. Since cocoa butter is very expensive, this reduces the cost of production of chocolate immensely.
Lecithin also helps in stabilizing
the dispersion and makes the chocolate resistant to temperature changes,
addition of moisture, agitation and storage. Many believe that it gives
chocolate better taste, gloss and increases its resistance to fat and sugar
bloom. Other surfactants have been proposed for the stabilization of texture
and retardation of bloom. However, lecithin remains unparalleled where
reduction in viscosity is concerned.1
The other surface-active agents used in the chocolate industry are ammonium phosphatides (YN) and polyglycerol polyricinoleate (PGPR). Ammonium phosphatides (YN) in dark chocolate has the same effect on the plastic viscosity but does not reduce the yield value as much as lecithin (126% of the value of lecithin). It increases the plastic viscosity (130% of the value of lecithin) in milk chocolates. The yield value is reduced to half the value for lecithin in milk chocolates. Polyglycerol polyricinoleate (PGPR) has significant effect on the yield values resulting in zero values (Newtonian flow) with 0.7% addition. It doubles the plastic viscosity in milk chocolates but has no effect in dark chocolates.2
Table V.a Flow characteristics of plain chocolate with added
surface-active lipids at 50oC3
![]()
Casson plastic Casson yield
Viscosity value
Addition (poise) (dynes/cm2)
![]()
![]()
![]()
0.3% soya lecithin 6.1 92
0.3% YN 10.3 30
0.3% sucrose dipalmitate 8.6 166
0.3% PGPR 32.5 25
![]() 0.8% PGPR 20.3 0
0.8% PGPR 20.3 0
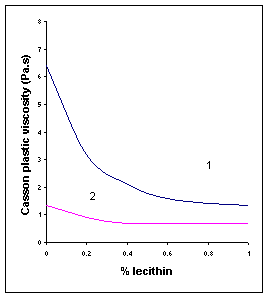
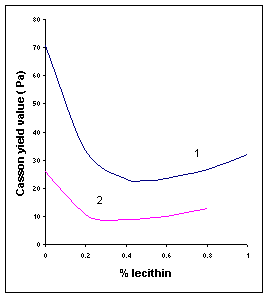
Figure V.a Influence of soya lecithin addition on Casson parameters of two dark chocolates. (1) 33.5% fat, 1.1% water; (2) 39.5% fat, 0.8% water4
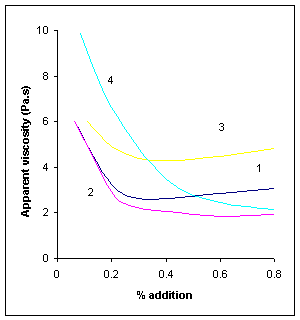
Figure V.b Viscosity reduction of dark chocolate by soya lecithin and by synthetic active lipids. Apparent viscosity determined at shear rate 15 s-1 and 50oC; initial apparent viscosity before addition: 19.5 Pa s or 195 poises. (1) Soya lecithin; (2) phospholipid YN; (3) sucrose dipalmitate; (4) polyglycerol polyricinoleate, PGPR5
Lecithin and its contribution in stabilizing emulsions6
Carbohydrates, proteins and fats, are the three main components of food. They are linked by various types of chemical and physical bonds. Surface-active emulsifiers such as lecithins are used to modify the interactions at the interface of dispersions and emulsions. Emulsifiers have hydrophilic (water soluble) parts and lipophilic (oil soluble) parts, and hence they concentrate in the interface between oil and water.
In food processing industries, lecithin is used for a mixture of phospholipids and oil, derived from oil seeds. Fig V.c6 gives the molecular structure if three main phospholipids in soy lecithin.
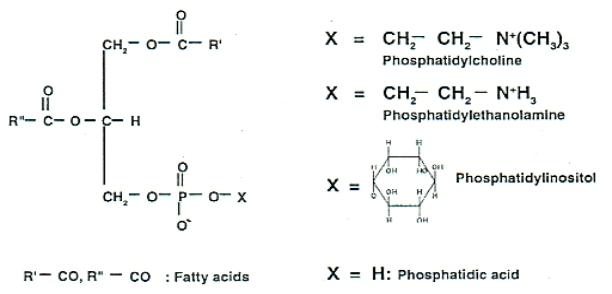
Fig V.c Molecular structure of main phospholipids
Basically, there are two types of emulsion: oil-in-water (o/w) and water in-oil (w/o) (Fig V.d7). Such emulsions are thermodynamically unstable and normally the emulsion would separate into two stable phases: oil and water. The destabilization and separation of emulsions usually occurs in four steps. (FIG V.e7)
Specific emulsifiers contribute to the reduction of the surface or interfacial tension. This is referred to as electrostatic stabilization. The food processor is aiming to supply the consumer with a stable food emulsion with excellent sensory and physical properties.
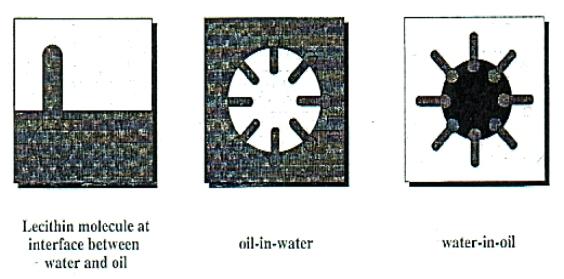
Fig V.d Types of emulsions: o/w and w/o emulsions
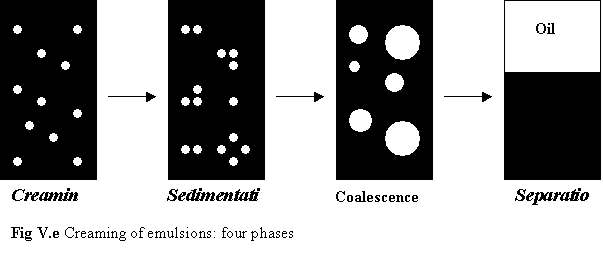
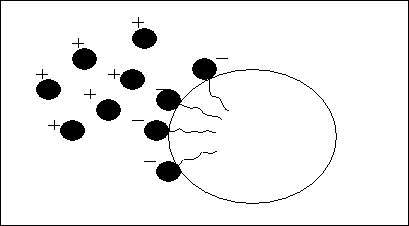
Fig V.f
Stabilization of emulsion
Phospholipids at the interface: there are different models
for the phospholipid structure at the interface. These can be seen in Fig
V.g8
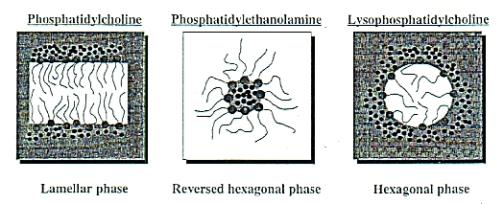
Fig V.g Phospholipid structure at the interface of an emulsion.
Chocolate is an emulsion with hydrophilic sugar particles and lipophillic cocoa particles. All of these are dispersed in cocoa butter which is a fatty medium. It has been shown that the interaction between lecithin and sugar is much more than the interaction with cocoa particles.
Mechanism of viscosity reduction9
Theory suggests several different but allied ideas to explain the action of lecithin
1. a monomolecular film is formed on the surface of the non-fatty particles by surfactant molecules.
2. reduction in internal friction by promoting the coating of sugar and cocoa solids by fatty medium.
3. increase in the amount of ‘free’ cocoa butter in the dispersion medium by displacement from the surface of solids.
4. prevention of agglomeration of sugar particles and cocoa particles by break down of any lattice-type structure between them.
5. reduction of non-fatty particles to smaller dimensions and hence the production of a more uniform dispersion.
6. absorption of moisture
There is a considerable degree of overlapping between the concepts put forward above and all except (5) would be expected to lead to a reduction in viscosity.
VI.
PACKAGING AND STORAGE1
It is very typical in the food industry that our eyes have to be satisfied first before our mouth. This is so true when it comes to the world of chocolate. The vibrant wrappers of chocolate not only attract the eye of costumers but it plays a very important role to present chocolate confectionery in engineering aspects.
As might be expected with a product, which has existed comparatively unchanged so many years, the basic packaging requirements of chocolate are relatively straightforward. It needs protection against handling dirt, insect infestation, taint as well as moisture. There are several forms of packaging in which chocolate confectionery in presented and packaged.
Molded chocolate
blocks:
Molded block packaging has been afforded by aluminum foil or tinfoil with a paper band for labeling product information. It takes up no extra space but within chocolate itself. Therefore, distribution costs are minimized. Besides, the over wrapper in molded block packaging provides wide appealing for legal information such as ingredients, expiration date and so on. It meets the requirement of maximum product display, which insist by many large customers.
Chocolate countlines:
The so-called countlines generally has irregular shape and enrobed rather than molded. However, it has been revolutionized by the advent of the “flow wrap”, “pillow pack” and “fin seal”. The machine takes a continuous web of flexible material and forms it into a tube, which wrap around product. Then, the edges will be sealed together by heat or pressure as it progresses through the machine. The tube containing the product is then again cut according to its length. They normally suffice for it to exclude moisture vapor, and potential taint.
Boxed chocolate:
The packaging of boxed chocolate is usually ranging from rigid handmade boxes with plinth bases, metal feet to machine-erected single walled cartons. Some materials such as flock paper, covered straw or chipboard, to metallic or film laminated carton board. A thin layer of greaseproof sheets or glassine commonly include as the inner fitments of chocolate boxes. It prevents crushing of the sweets as well as cushioning. For some nutty sweet such as paralines or coconut clusters exude fat to such an extent that they can pervade glassine or cellulose layers. Therefore, they required a sheet of impermeable film like plastics to prevent staining of the box itself.
When boxes of chocolate are combined together in an outer, an important decision needs to be made whether they to should travel flat or on edge. It is often better to pack the carton on edge. So that, the thermoformed tray can relieve the pressure, provided it is strong enough. In a conclusion, the construction of outers and cases is much similar for bar and countlines.
Twist wrapping:
Twist wrapping of chocolate packaging has been a major part of the confectionery scene for years. The materials used are aluminum foil, backed or unbacked plain sliver or colored, and film, tinted, clear or printed. The most general form of twist wrapping is the “double end fantail”. Two ends of the wrapper will twist and turn into shape to enfold the chocolate. Twist wrapping usually comes with varies type of colors and materials which attract most of the consumers. They also lend themselves to packaging in printed film bags, generally with display windows, produced on vertical form-fill-seal machines. Materials such as Polyproplene and polyethylene present in one form or another form often use in making the bags. Such bags are intentionally to be hung for display purposes.
The technology behinds chocolate packaging is complex. Determination for the used of material is crucial. It has to compromise the characteristics of chocolate that it does not melt until it is put in the mouth. In addition, the materials should be capable protect against moisture vapor, taint and dirt. There are several types of materials, which are widely used in today market.
Aluminum foil:
According to the chocolate manufacturer, aluminum foil is the best barrier to water vapor and gas transmission which available in flexible form. For commercial purposes foil thickness can range from 7-12 um. Recently, cost reduction has taken the form of the special alloys, which is the combination of aluminum and manganese. It provides extra strength compare to pure aluminum. For example, 8 um material can now be obtained that is stronger than the old 9 or 10 um foil. Some materials such as cellulose and plastics film are used for extra barrier properties and puncture resistance. When foil is coated, usually with vinyl or polyethylene in order to make it heat sealing. Both of them are beneficial to chocolate confectionery.
Regenerated cellulose film (RCF):
Until relatively recently RCF dominated the flexible packaging field. In its natural state RCF has few characteristics properties other than transparency, flexibility and the ability to form a barrier to oxygen, provided it is dry. Besides, it is very sensitive to moisture, expanding or contracting with changes in atmosphere humidity. Therefore, to make RCF a useful proposition, it has to be coated or other treatments. Whenever extra protection is required, material such as PVDC (polyvinylidene chloride) is applied. Coated RCF is an ideal film for automatic packaging, as it is not subject to structural alteration because of temperature; however, it cannot be thermoformed. It offers combination of rigid with elasticity, it also avoids static electricity problem. Moreover,
Its tensile and burst strength are good. It will propagate easily once a tear was made. So, it is actually an advantage when a tear strip is required.
Plastics films:
Despites all its properties, RCF film has largely been supplanted by plastics films for flexible packaging. PVDC used to coat RCF, is itself a plastic and can be obtained in film form as well as coating. The range of plastic is very wide in world of chocolate. It plays an important role in presenting chocolate confectionery.
There are few plastic materials exist in packaging of chocolate for many years. There are polyethylene, polyvinyl chloride and polyester. Polyvinyl chloride or PVC is the most commonly used where clarity and sparkle are required in a film, rather than strength. For example, for shrink boxes wrapping method, it has good resistance to oils and fats. PVC also used in thermoformed trays for packing chocolate assortments. It will minimize the labor cost compare to the old glassine cups where both of them have equal functions. It has proven by the development of robotic packing.
Besides PVC, the other plastic film, which has achieved relatively widespread acceptance in confectionery packaging, is polyester, which is very strong and clear. As a packaging material is it probably most widely used in its metallic form. It enhances not only its appearance, but also its barrier properties against moisture, gas and ultraviolet. Those are the minimum necessity for quality assurance of chocolate industry.
To assure the quality of chocolate in a production line would hardly be possible without full electronic control of what is basically a series of mechanical operations. It can involve a multiplicity of tests to be carried out before materials reach the production line. Therefore, a well-equipped packaging should be aware of seal strength, tensile strength, protection against moisture, taint prevention and so on. For example, chocolate is particularly sensitive to external taint. Most modern flexible material are at least fairly good barrier to taint, and can be made very good by coating with PVDC or laminating. Besides, the quality seal is important: where package is not sealed, external taint sources must be eliminated. Incorrectly applied coatings or laminants such as polythene, oxidation of printing inks, varnishes can offset on to the inside of reeled. Therefore, manufacturer of chocolate should take every wary step to meet costumer demand on the irresistible, delicious chocolate.
VII. Marketing
The reason why chocolate is eaten world wide is because people find pleasure in it. Through taste and texture there is probably no other product that gives so much enjoyment to so many individuals. Chocolate and sweets bring color and excitement into everyday lives, and throughout adult life continue to symbolize gaiety, festivity and goodwill. Sweets are fun, and the range of products and prices is such that its enjoyment is within the reach of almost everybody.
Nearly all the population has eaten chocolate or confectionery at some time and over 90% may buy with regularity. Confectionery in this paper is referring to any product that contains cocoa beans, milk, and fats. Chocolate consumption is very high in many industrialized countries. United States itself consumes three billion pounds per year. In the United Kingdom, chocolate and chocolate confectionery accounts for up to 60 % by weight for the current UK market. These energy-rich foods are still increasing in demands due the convenience of these snacks. Chocolates, in the blocks or bar form, do not need preparation and may be eaten during a rush, or on the way to a certain place. Since these foods are very high in energy many people consume as a take-away snack.
The scale of production varies on countries. Some countries produce high-priced chocolates in the kitchen behind retail shops. In other countries, chocolates are being produced in mass production. These mass production facilities are spreading from the Western Europe and North America to countries in South East Asia, South America, and in the Middle East. Due to this vast production technology spreading, the Third World countries are benefiting. All the ingredients are exported from tropical countries. For example, 34% of all exports for cocoa are from the Ivory Coast, and sugar is from countries like Fiji, Mauritius, the West Indian Islands, and Cuba.
Chocolates will not survive in the market if it is not formulated well. The product must be designed, or re-designed from time to time. Certain factors must be taken into account in designing a chocolate. First of all, the primary function of the chocolate has to be met. Chocolate has to be more energy giving than just sweet taste in a low volume. The size of the product has to be decided. It can’t have too much volume and if it has too little, then people will consider it simply “a rip off”. The price has to be adjusted so it is not overpriced. The price has to be similar to the other competitors or else, the product will not sell. The efficiency of packaging in transit and labeling has to be put in factor too. The goal is so that people doesn’t have to hassle to open the package for eating. At the same time, it must be packaged well so that it will not make any mess when eating, and it has to have a decent amount of shelf life. Finally, some legal requirement has to be met. Every product in the market has patents so that other competitors won’t use the technology, and a violation of this should be avoided.
Despite all these designing of the chocolate, advertising is also a necessity in keeping the product on the market. Some forms of advertisement are in TV commercials, radios, magazines, and billboard posters. Larger companies, has enough money to have commercials for the product, whereas smaller companies can afford radios or magazines.
VIII. Reference Cited
1. Woodman, A.G., “Food Analysis”, 4th Ed, McGraw-Hill Book Company © 1941
2. Zipperer, “The Manufacture of Chocolate and Other Cocoa Preparations” (1915)
3. Hilditch, T.P., “Chemical Constitution of natural Fat”, 1956
4. Walstra, P & Jenness, R., “Dairy Chemistry and Physics”, 1984, John Wiley, New York
5. Augustin, M.A, “Dairy ingredients in chocolate- chemistry and ingredient interactions”
6. Reimerdes, EH & Mehrens, H-A,. “Milk In Beckett”, ST. (Ed). “Industrial Chocolate Manufacture and Use” 2nd ed., Blackie Academic & Professional, Glasgow; ©1994
7. Fox, B.A, Cameron, A.G, “Food Science”, 2nd Ed, University of London Press Ltd © 1970
8. Potter,N.N; “Confectionery and Chocolate Products”; Food Science; 4th edition; The AVI Publishing Company;
9. Beckett,S.T.; “Milling, Mixing and tempering-an engineering view of chocolate”; Proceedings of the Institution of mechanical Engineers. Part E; Journal of process mechanical engineering; 2001; 215(E1);1-8
10. Chevalley,J.; Chocolate flow properties; Industrial Chocolate Manufacture and Use; 2nd Edition; Blackie Academic & Professional
11. Aylward,F.; “Chocolate”; Texture in Foods; SCI Monograph(7); Society of Chemical Industry; ©1960
12. Ley,D.; Conching; Industrial Chocolate Manufacture and Use; 2nd Edition; pg.122; Blackie Academic & Professional
13. Niewwenhuyzen, W.; Szuhaj.B.F; “Effects of lecithin and proteins on the stability of emulsions”; Fett-Lipid; 100(7); Jul 1998
14. Martin A.V., Ferguson N., “Packaging”; “ Industrial chocolate manufacture and use ”; second edition, BLACKIE ACADEMIC AND PROFESSIONAL © 1994