Phthalate
Plasticizers
CE 435
April 6, 2001
Group:
Brian
Amato
Sarah
Karl
Carla
Ng
Contents
Theory: Mechanism of Plasticizer
Effect on the Polymer
The Phthalate Plasticizer Market
Health Issues in the Use of
Phthalate Plasticizers: Are They Safe?
Plasticizers: An Introduction
A plasticizer is a polymer additive that serves to increase the polymer’s flexibility, elongation or ease of processing (workability). In more technical terms, the addition of a plasticizer generally causes a reduction in the cohesive intermolecular forces along the polymer chains. The chains can then move more freely relative to one another, and the stiffness of the polymer is reduced.
Plasticizers are usually inert organic materials with high boiling points and low vapor pressures. Esters are commonly used due to their favorable physical interactions with high-molecular-weight polymers. This physical interaction causes the polymer and plasticizer to form a “homogenous physical unit.” In other words, they do no separate out. Two classes of plasticizer exist: primary and secondary. Primary plasticizers are, in a way, “true plasticizers.” These are the chemicals that interact with the polymer to increase its flexibility. Secondary plasticizers, on the other hand, act not on the polymer but the primary plasticizer, increasing its effectiveness (10).
There are two primary means of plasticization: internal and external. Internal plasticization involves the chemical alteration of a polymer or its monomer (prior to polymerization). This can be done either by random copolymerization or side chain grafting. The former tends to increase the flexibility (aided by the random quality; a more ordered copolymerization would imbue a higher degree of crystallinity thereby giving the opposite effect). The latter lowers the glass transition temperature, Tg, and reduces crystallinity by disrupting physical interactions between the chains.
The second type of plasticization is external. It is what we normally think of when we talk about a plasticized polymer, and involves the addition of an organic chemical (see above) during compounding (after polymerization). This chemical interacts with the polymer only physically, via its solvent capabilities. This type of plasticization is usually done at elevated temperatures.
External ester plasticizers are the most commonly used in the industry. They carry the advantage of great flexibility: since they are added after polymerization plastics of varying degrees of flexibility may be produced from one polymer formulation.
Early Plasticizers
The use of plasticizers began in the mid 1860’s when castor oil was added to Cellulose Nitrate (CN). The most commonly measured physical effects include melt viscosity, elastic modulus, and glass transition. Other types of plasticizers have been used through the years, but in the late 1930’s phthalates were introduced because they were believed to be less toxic. The two most common types used today are di-2-ethylhexyl phthalate (DEHP) and di-isononyl phthalate, which are generally used in PVC related products. In today’s market many medical delivery systems, children’s toys, and baby devices are made of PVC. Recently there has been increasing debate about the danger of phthalates leaching out of the PVC and into individuals. It is still unclear what dangers these chemicals pose to humans, but research on a variety of other species has been conducted for quite a while and has shown mixed results.
Theory: Mechanism of Plasticizer Effect on the Polymer
There are several theories that seek to explain how a plasticizer affects a polymer in both internal and external plasticization. Three major theories are described below, as well as some important additions to the theory (10).
i) The Lubricity Theory – this theory describes the effect of an external plasticizer on a polymer in terms of lubrication. A “dry” polymer, a resin without plasticizer, is rigid because friction exists between its chains, binding them into a network. When the polymer is heated in order to be plasticized, the binding is weakened and the smaller plasticizer molecules are able to slip in between the chains. When the polymer cools, the plasticizer molecules act as a lubricant between the chains, allowing them to “slip.”
ii) The Gel Theory – an extension of the first theory, the Gel theory proposes that the plasticizer molecules break up the polymer-polymer interaction by getting in between the chains and “obscuring” these interaction sites from the polymer molecules.
iii) The Free-Volume Theory – this more expanded theory allows for some quantitative analysis of polymer-plasticizer interaction. The free volume of a polymer can be described as the “empty internal space” available for the movement of the polymer chains. It has been shown that the free volume of a polymer greatly increases when it reaches the glass transition temperature. By this logic, then, the study of plasticization is essentially the study of methods for lowering the glass transition temperature. We know that the glass transition temperature is the point at which significant, one might say concerted, molecular motion begins to occur. This motion, which corresponds to an increase in the free volume of the polymer (which is what we want), can be due to motion of the chain itself, of its ends, or of side chains attached to it. So how does the addition of the plasticizer facilitate this? Its lower molecular weight means an increase in the free volume per volume of material (you are not adding a lot of volume with these small molecules, but there is a lot more free space between them than between the polymer molecules). If you are using a small polymer molecule as a plasticizer, it has a lower glass transition temperature than the resin you are adding it to, so the Tg of the resulting mixture will be lower.
So the addition of a plasticizer in essence allows a polymer to behave, at room temperature, in a way the pure resin would only behave at elevated temperatures.
Phthalate Plasticizers
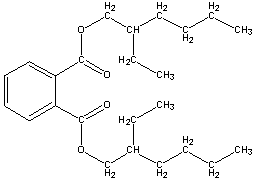
The specific type of plasticizer that is of concern to this report is the phthalate plasticizer. Phthalate plasticizers are the most commonly used plasticizers in PVC. The PVC polymer chains are attracted to one another, and therefore from a very rigid structure. The phthalate plasticizers are added to PVC to help the chains slide against each other, therefore, softening the PVC. The structure of phthalate plasticizers is that of a phthalate ester, which is simply a phthalate with, an ester group (see structure below figure 1) (1).
(Figure 1.)
Phthalate plasticizers are colorless liquids like vegetable oil with a faint odor, and they are insoluble in water. They are however, miscible in mineral oil, hexane, and most organic solvents. This makes them readily soluble in bodily fluids, such as plasma and saliva (1).
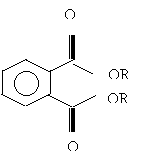
Two good examples of phthalate plasticizers are DEHP ( Di-Ethylhexyl Phthalate), and DINP (Di-Isononyl Phthalate). DEHP has been the most commonly used, and is still the plasticizer of choice for all PVC medical and surgical products. However due to evidence of the toxicity of DEHP in laboratory animal studies it was replaced in children’s products with DINP. The structures of both are shown below in figures 2 and 3 respectively (1).
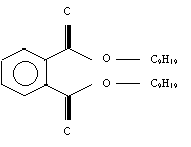
(Figure 2. DINP-Chemical Structure)
(Figure 3. DEHP- Chemical Structure)
The Phthalate Plasticizer Market
In 1999, the
global volume of plasticizers was approximate 10 billion lbs, or about $5
billion. The market has an average
yearly growth rate of 2-3%.
Of the ester plasticizers, standard phthalate esters comprise over 85% or the tonnage produced every year. They command the market due to their low cost and easy availability. Other common plasticizers include specialty phthalate esters, adipates, and trimellitates (which are used for low-temperature applications).
Over 90% of the plasticizer volume produced every year goes into Poly(Vinyl Chloride), or PVC. This polymer is found in anything from food packaging to construction materials to toys to medical devices. Yet PVC in its “true” form is a hard, brittle polymer. Without the existence of plasticizers, it would have very little commercial use. It is a symbiotic relationship, however: few polymers interact as favorably with plasticizers as PVC. Not only is it capable of taking in high concentrations of plasticizer, it also retains them much better than most polymers. This is partly due to the very heterogeneous morphology of PVC. It contains regions that are highly amorphous, semi-crystalline, and highly crystalline.
In choosing an appropriate plasticizer, several considerations are important. One must take into account what the application of the polymer will be: will it be high-temperature or low-temperature? What degree of flexibility is needed? The size of the plasticizer molecule, and more importantly its bulk (taking into account the presence of branches or side chains) will affect both the efficiency and the temperature behavior of the polymer. Efficiency in this case is basically a measure of how much plasticizer must be added to achieve a give amount of softness in the polymer. The larger the molecule (more carbon atoms), the lower the efficiency. More highly branched molecules also make for less efficient plasticizer. However, increasing the size of the molecule increases its mass, and therefore lowers volatility. This is desirable if your polymer is to be used in high-temperature applications, where more volatile molecules would evaporate out. On the other hand, if your polymer is to be used in low-temperature applications, such as flexible tarps for use outdoors, highly linear plasticizer molecules are desirable.
For most common household applications that use PVC, the polymer is not being subjected to any temperature extremes, aside from the somewhat high temperate of the water which hits your plasticized PVC shower curtain every day. For this reason, the “mid-range”-sized phthalate esters, which have the benefit of being inexpensive, are a good choice.
Figures 4 (a) and (b), below, show two common applications of plasticized PVC.
Figure 5 shows PVC without the use of plasticizers—common PVC piping, which is not flexible at all! It’s quite surprising to think that these three very different materials are all made from the same resin.

Figure 4: (a)
rubber ducky and (b) blood bag, both made from plasticized poly(vinyl
chloride).
Figure 5: PVC pipe, made without the use of
plasticizers, is a stiff, strong material.

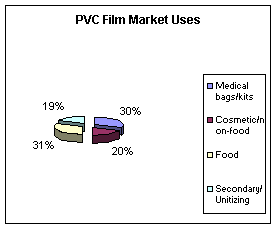
Figure 6, below, shows some market data for the use of PVC (3).
Figure 5:
PVC use in the U.S. market.

Health Issues in the Use of Phthalate Plasticizers: Are They Safe?
Phthalate plasticizers have been found to be a health concern when found in direct contact with bodily fluids. Studies performed on laboratory animals have shown that there is direct evidence that certain phthalate plasticizers have a carcinogenic effect in vivo. Because they are readily miscible in organic solvents like plasma and saliva, humans have a chance of ingesting or absorbing them during common medical procedures. It is believed that once they are absorbed they are stored in the fatty tissue of humans, and therefore can be teratogenic. There is also little known about the body’s ability to metabolize them once they are ingested or absorbed.
Types of procedures where there is a risk of absorption or ingestion occurring are during blood transfusion, use of tubing for respiration, or in the use of catheters. When PVC is used for blood bags the plasticizer can make up from 20-30% of the final weight (2). Since the plasticizer is not covalently bound to the PVC molecule it can diffuse out into the plasma. When DEHP is dissolved in plasma it is a result of the miscibility in albumin or lipoprotein.
The choice of PVC for storing blood came about due to the increased survival rate of red blood cells when stored in PVC as compared to glass (2). Research in the 70’s at John Hopkins concluded that DEHP from the PVC bags contaminated the stored blood 24hrs after filling the bag to a concentration of 2.5 milligrams of DEHP per liter of blood (2). It was concluded from this that an adult could get a dose from a bag of this type of nearly 300 milligrams total or 5 milligrams per kilogram, and for a child it would be even higher (2).
Since these studies, which show that DEHP is potentially harmful, there have been more recent studies that show that it is actually beneficial to the production of cholesterol and phospholipid production during storage. From this study a dose response curve was made which shows that the greater the concentration of DEHP to which the red blood cells were exposed, the lower the hemolysis (death of red blood cells) during storage. As to what exactly causes this phenomenon, there has never been a definitive answer. It may be that the plasticizer is some how interacting with the red blood cell cytoskeleton to neutralize any effects of oxidation or disconnection of the cytoskeleton, which would also lead to increased micro vascular formation (2).
From all of the research to date on this issue the consensus of the FDA is that further investigation needs to take place into the reproductive toxicity of DEHP. Also alternative plasticizers need to be investigated, and their toxicity needs to be evaluated in turn.
A commonly asked question is, what are the consequences of using these chemicals? This is not a dilemma just for the plastics industry or Green Peace, but rather every person and creature on the earth whose lives are impacted by the use of plastics. It is a truth that plastics have taken over many roles in the world we live in today, to a point where it is impossible to escape them. From the containers of the food we eat, the trash we produce, to the pacifier that we give to a child to teethe on. Plastics are unavoidable. What are the dangers of phthalates if there are any and how will they affect the overall environment? Currently there are limited studies that quantify the danger of phthalates to humans, but the effects demonstrated on some species are undeniable.
Evidence?
Research with laboratory rats has correlated DEHP with malformations in male rats by decreasing fetal testosterone levels. This indicates the possibility of transgenerational reproductive toxicity. A study done by the Environmental Protection Agency’s Reproductive Toxicology Division shows fetal testosterone levels in male rats similar to those of female levels at a critical stage of reproductive development when the mother was treated with 750 mg/kg/day from gestation day 14 to postnatal day 3 (Toxicological Sciences 58). Evidence similar to that above is prevalent, but how does it relate to humans? Well it doesn’t necessarily. Many researchers are considering the possibility of species specificity concerning the effects of phthalates. While the idea of a chemical behaving so detrimentally to one species and yet have little to no effect on another seems peculiar, a study on Cynomolgus Monkeys clearly supports this notion. This study is quite interesting because monkeys are primates as are humans. Therefore many of their biological processes are similar if not identical. With that in mind tests were performed using DEHP and DINP at 500 mg/kg/day for 14 days by intragastric intubation. Histopathological examination of tissues showed no distinctive treatment-related effects to the liver, kidney, or testes unlike that in tests performed with laboratory rats(Toxicological Sciences 56). Also, there were no overt changes in animal behavior as a result of the treatment and no effect on body or organ weight was documented. While this does not give phthalates a green light it does show that some species are not good representations of the effects that these chemicals have on all species including humans (Toxicological Sciences 56).
There are still incidents with humans that are cause for concern with the use of phthalates. Young girls in Puerto Rico are a prime example. For quite some time now there has been an inexplicable epidemic of premature breast development, also know as thelarche in girls between the ages of 6 to 24 months (Science News, Sept. 9). It strikes about 8 out of every 1000 and is currently the highest known rate. Initially it was believed to be the result of pesticide use. Yet a study at the University of Puerto Rico, San Juan examining the blood work of a group of girls suffering from thelarche showed no signs of pesticides, but rather phthalates. This was finding was surprising, but after some reflection seemed logical. Since Puerto Rico is an island it must import much of the food it consumes. Packaging for food is generally made of plastic that contains phthalates. Another factor that may be contributing to the effects of phthalate leaching is Puerto Rico’s tropical climate. The high temperatures increase instability of plastics and higher rates of leaching of phthalates are observed. A similar test including 41 girls suffering from thelarche and 35 developing normally was conducted in San Juan. 68% of the girls suffering from the condition had detectable levels of phthalates in their system compared to only 17% of the girls developing normally (Science News, Sept. 9). This study is inconclusive, but nonetheless it shows that more studies need to be performed to determine the effects that these chemicals have on individuals.
What’s
Being Done In The Meantime?
So no one knows for sure what effects phthalates have on individuals, but what is being done until more information is available? Should the government get involved and ban the use of these chemicals? Industry argues that such an action would be premature and there isn’t enough evidence present. Even so, out of good faith many manufacturers have chosen to be cautious and stop use of these phthalates. A list of companies who have pledged to remove phthalates from many of their products includes Disney, Evenflo, Safety First, The First Years, Gerber, Sassy, Shelcore Toys, Hasbro, and Matel (Mothering March 1999). But what about those companies who haven’t made these promises to the public? Groups like Green Peace have suggested caution on behalf of the consumer. Take the time to educate yourself and know what you are purchasing for both you and your family. If you aren’t sure what a product is made of, call the manufacturer and ask. Manufactures are required to inform you what a product is comprised of. In this situation, ignorance may prove detrimental. In turn taking some initiative and could be invaluable to your health.
Health or Hype?
It can be difficult to decide, when it comes to industry
vs. consumer interest groups, what to believe.
All too often the number one concern of industry is the bottom line, and
similarly consumer interest groups have some very different agendas from what
they advertise.
It is interesting to note that phthalate plasticizers first entered the market as a safer alternative to thioesters. They have been in use for close to fifty years and, because of their applications in plastics used in the medical industry, have been thoroughly studied. However, the rate of plasticizer elution from a polymer is hard to measure consistently and reliably, and therefore there is no way to gather conclusive results as to the amount of phthalates we may be continuously exposed to from countless sources.
Groups like GreenPeace have gained reputations as being somewhat extremist. Their campaigns against potential environmental and health threats often smack of propaganda, and one feels the need to take their allegations with a grain of salt. In a well-written defense of the vinyl industry (and, perhaps more accurately, an attack against GreenPeace and its tactics), Bill Durodie of the Competitive Enterprise Institute (6) brings up some persuasive evidence that GreenPeace’s claims are overblown. He concentrates on their campaign to ban the use of PVC, and the phthalates that are used with it (specifically DEHP and DINP), in children’s toys.
Of main concern are PVC teething rings, which are of necessity continuously exposed to a child’s mouth and their saliva, which can act as a solvent for the plasticizer.
Durodie notes that many of the “research” papers used as evidence by GreenPeace were actually examples of bad science and poorly run experiments, or good science misinterpreted. His main concern was that the recent trend, particularly in Europe, was to discredit the evidence found by science in favor of unfounded, reactionary concerns. This is relevant not only to the plasticizer issues, but also to things like genetically modified foods, for which we’ve seen much of a similar reaction in Europe. Of course, his position is similarly worth examining with skepticism, as his paper is not without traces of propaganda-ism itself. We are left then, to our own devices.
Many of the objections to DEHP and DINP are based on their being peroxisome-proliferating compounds. These are compounds that cause mutation and spontaneous growth of cells. In particular, they are thought to be hepato-carcinogenic, meaning they cause liver cancer. Although a number of studies have shown this to be true in rats, newer studies are showing that this may not be relevant to us, due to the fact that primate cells do not show the same reaction to these compounds (12)(13). These recent studies seem to suggest phthalates are benign chemicals, safe for human use. As was discussed earlier, though, other studies such as the one in Puerto Rico imply there may still be cause for concern.
The safest conclusion to be drawn from this is that it would be to the detriment of scientific advancement to always doubt science, yet at the same time disregarding possible health concerns could lead to trouble down the line. In the end, “Buyer Beware” may be a little extreme, but “Buyer be Informed” is good advice.
References
- Abbott, Barbara D. (2000). “The Plasticizer Diethylhexyl Phthalate Induces
Malformations by Decreasing Fetal Testosterone Synthesis during Sexual
Differentiation in the Male Rat.” Toxicological Sciences 58, 339-349
- Ackley, David C. (2000). “Effects of Di-isononyl Phthalate, Di-2-ethylhexyl
Phthalate, and Clofibrate in Cynomologus Monkeys.” Toxicological Sciences 56,
181-188
- Carraher, Charles E. Polymer Chemistry: an Introduction 4th Ed. (1996) Marcel Dekker, Inc. NY, New York 424-426
- Durodie, Bill “Poisonous Propaganda, Global Echoes of an Anti-Vinyl Agenda.” Competitive Enterprise Institute, July 2000
- Christensen, Jackie Hunt. Toxic Toy Story. Mothering. Sept. 1998 p38 (1)
- O’Mara, Peggy. Winning the Fight Against PVC. Mothering. March 1999 p35 (1)
- Raloff, J. Girls May Face Risks from Phthalates. Science News. Sept 9, 2000 v158 ill p165
- Ullman’s Encyclopedia of Industrial Chemistry, Vol A20 pp439-451, VCH Publishers, Inc, 1992
- Woodyatt, K.G. Lambe, K.A. Myers, J.D. Tugwood and R.A. Roberts, “The Peroxisome Proliferator (PP) response element upstream of the human CoA oxidase gene is inactive among a sample human population: significance for species differences in response to PPs.” Carcinogenesis, vol.20 no.3 pp.369-371, 1999
- Zacharewski, T.R., M.D. Meek, J.H. Clemons, Z.F. Wu, M.R. Fielden and J.B. Matthews, “Examination of the in vitro and in vivo estrogenic activities of eight commercial phthalate esters.” Toxicological Sciences, Vol. 46, pp.282-293, 1998