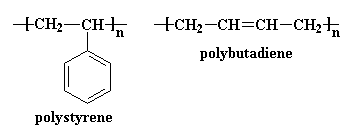Plastics in the Microwave Oven
CE 435

(Photo courtesy
of Reynolds Metal Company)
Submitted
to:
Dr. P. Alexandridis
Assistant Professor of Chemical Engineering
State University of New York at Buffalo
Submitted
by:
Daniel
Fulcher
Christopher
Hunter
Martin
Schaefer
April
6, 2001
Table of Contents
Home Meal Replacement Packaging
Introduction
During the 1980s, the general public’s growing understanding of the safety of microwave ovens resulted in the number of U.S. households owning a microwave oven to increase from 15% in 1980 to 78% by 1989.1 For this reason, the demand for food products designed for microwave ovens grew, thus spawning the creation of a new line of food products. According to Gorman’s New Product News report, the number of new microwave product introductions increased from 278 in 1986 to almost 1000 in 1988.
Additionally, the consumer desired everyday cookware and storage containers that were safe to use in the microwave oven. Thus, Tupperware®, Rubbermaid®, and others redesigned their plastic storage containers for use in the microwave oven. These products provided a lightweight, inexpensive alterative to glass containers. Further, the generally reduced cooking times in microwave ovens tend to retain more healthy vitamins than those cooked traditionally.2
Microwave ovens heat water molecules in food by causing the polar molecules to fluctuate at a specific frequency. The motion of the water molecules relative to each other creates frictional heat. A common misconception is that microwaves in microwave ovens excite a natural resonance in water. However, the frequency of microwave ovens is ~2.45 GHz, which is well below the natural resonance on an individual water molecule. The choice of ~2.45 GHz was chosen to ensure effective penetration into the food and stimulation of water molecules to generate even heating throughout the food.
Consumer desire for quick, convenient, and high quality food preparation has created an industry with combined approximate annual revenues of 3 billion dollars. The food industry has progressed from packaging traditional foods, to engineering foods and packaging specifically for use in microwave ovens. However, many food manufacturers package frozen meals in containers that can be heated in both conventional and microwave ovens, to prevent exclusion of the conventional oven market share.
Scope
Heat resistance and mechanical strength are essential physical properties for polymer containers used in microwave ovens. Polymer containers must withstand temperatures up to ≈ 230oF for microwave ovens and up to 400oF for dual oven applications. Additionally, the containers should be sturdy, rigid in shape, and capable of supporting its contents before and after heating in the microwave.
Packaging and storage polymers are colored specifically for the intended application. Generally, food packaging is a nontransparent color to prevent ultraviolet light from interacting with the food. In contrast, short-term consumer food storage containers are transparent, so the contents are easily identified.
The properties of pure polymer resins are frequently altered by additives, such as fillers, colorants, and plasticizers. These additives can change both the physical and chemical properties of the polymer, thus giving the desired characteristics needed for cooking applications.
The electromagnetic radiation used in microwave ovens does not directly interact with polymer containers. However, food products that contain oils and fats can create localized temperatures of ≈ 300oF on the surface of the container. This localized heating may result in polymer degradation or conformation changes, resulting in migration of polymer contents into the food. Thus, testing is necessary to ensure the containers do not pose any potential health hazards.
The Food and Drug Administration’s (FDA) establishes regulations and testing standards for the food container industries. The manufacturer’s test results must show the nature and levels of the packaging material, the components used and the possibility of migration, and any potential health hazards.3 In addition, the FDA tests food contact surfaces for migration of polymers, and polymer additives into food. FDA approval of a product ensures that the product is safe for use as a food contact surface, and it complies with all standards of industry.13
Home Meal Replacement Packaging
Home meal replacement (HMR) packaging can be split into two general categories: microwave oven only and dual oven (conventional and microwave). A common polymer used for dual oven packaging is crystallized polyethylene terephthalate (CPET), while microwave only packaging is usually made of polyphenylene oxide high impact polystyrene blend (PPO/HIPS). However, both categories can use the same coverings, since the cover is removed when cooked in the conventional oven. A typical HMR container is presented in figure 1.

Figure 1: Food Tray and Covering8
CPET
Chain Structure and Morphology
PET is a heterochain, modified homopolymer, which is created by the step growth condensation polymerization reaction of terephthalic acid and ethylene glycol. The chain structure of PET monomer is presented in figure 2.
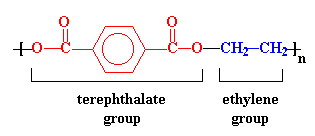
Figure 2: PET Repeating Unit10
Organic or inert nucleating agents
are added to allow PET crystallization to occur during the extrusion
process. A target crystallinity of
approximately 30% is desired for semi-crystalline CPET dual oven trays.
Properties
HMR dual oven packaging requires an operating temperature range of -40°F to 400°F. Crystallization of PET increases the upper temperature resistance from 230°F to 400°F, which provides dual oven capability. However, CPET may become brittle at low temperatures, so a balance is required between strength properties and temperature resistance. For this reason, manufacturers produce trays with a crystallinity of 28%-32% and an intrinsic viscosity of .85 to .95.4 Additionally, some manufacturers produce trays with a two-layer structure of CPET for rigidity and temperature resistance and amorphous PET for impact strength at low temperatures.
CPET
provides good gaseous barrier properties against both oxygen and carbon
dioxide. Further, CPET has acceptable
water barrier properties for HMR food
applications.
The crystalline structure of CPET
alters the normally transparent PET to an opaque color. However, CPET is easily colored with a
pigment. The industry standard is
black, using black carbon as a pigment additive. Additionally, the nontransparent packaging prevents ultraviolet
light interactions with the food.
Processing
HMR trays are normally produced using a thermoforming process. One specific method of thermoforming CPET is Vacuum Snap-Back Thermoforming. Figure 3 diagrams the basic thermoforming process.
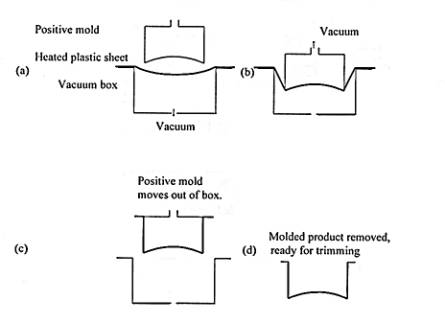
Figure 3: Vacuum Snap-Back Thermoforming6
The heated sheet is pre-stretched into the vacuum box before forming with the positive mold. The pre-stretching creates a more uniform wall distribution, which allows this process to create deep-dish trays.6 The final CPET trays have a high gloss and a hard surface.
In 2000, 84 million pounds of CPET trays were produced at an approximate cost of .52 $/lb.5,12 Eastman Chemical Company produces one specific form of CPET resin under the product name of VersaTray®.
PPO/HIPS
Chain Structure and Morphology
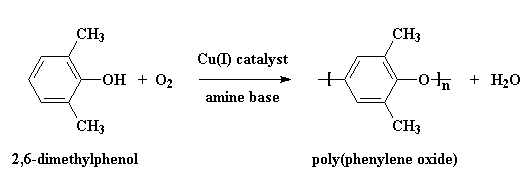
PPO/HIPS is a fully miscible, blended polymer developed and manufactured under the name Noryl® by General Electric Plastics. PPO is a heterochain homopolymer, which is created by a free-radical, step-growth, oxidative-coupling polymerization of 2,6-dimethylphenol and oxygen. The polymerization reaction is presented in figure 4.
Figure
4: PPO Polymerization10
HIPS is a grafted copolymer of cis-1,4-poly(1,3-butadiene) and styrene monomer. The double bonds in the polybutadiene allow the graft polymerization to occur when polymerized with styrene monomer. The resulting polystyrene chains are atactic. The chain structures of HIPS repeating units are presented in figure 5.
Figure 5: HIPS Repeating Units10
Typically, HIPS blends contain between 2% to 15% by weight of polybutadiene to impart the desired impact properties.6 Figure 6 presents a simplification of the polymer structure.
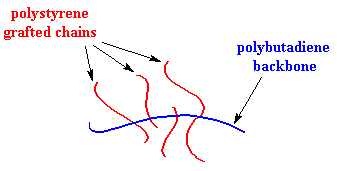
Figure
6: HIPS Graft Polymer Structure10
Even though a copolymer is formed, polystyrene (PS) and polybutadiene are immiscible and phase separate creating compartmentalized regions within the grafted polymer. Since polybutadiene is the minor component of the polymer, the polybutadiene forms small, dispersed globules within the polymer structure.
The Noryl® PPO/HIPS blend is approximately 25% PPO and 75% HIPS by weight and results in an amorphous polymer.
Properties
Mixtures of PPO/HIPS are fully miscible and improvements in the final properties are in direct proportion to the amount of PPO added.5 PPO is added to HIPS to increase temperature resistance from approximately 180°F to 230°F, for use in the microwave oven.
PPO has a low resin flow and is difficult to form, so blending with HIPS improves flow characteristics and allows for thermoforming on equipment designed for PS.4 Additionally, the small, dispersed globules of polybutadiene elastomer within the PS phase absorb impact energy, thus improving the PPO/HIPS blend impact strength.
The poor gaseous and water vapor barrier properties of HIPS are greatly improved by PPO, thus making the final blend acceptable for HMR packaging.
The PPO/HIPS blend results in an opaque color. However, the blend is easily colored with a pigment. Like CPET, the industry standard is black, and manufacturers use black carbon as a pigment additive.
Processing
To ensure accurate blending during the extrusion process a high intensity mixer is required.5 One specific method used for thermoforming PPO/HIPS is the Thermoform-Fill-Seal process. Figure 7 diagrams the basic thermoform-fill-seal.
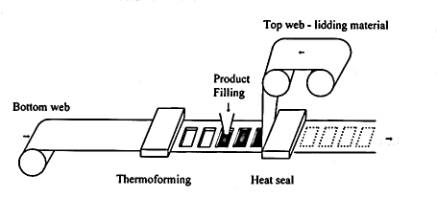
Figure 7:
Thermoform-Fill-Seal6
Coverings
Most companies in the microwavable food industry heat-seal the food containers with a multi-layer polypropylene film. Companies stopped using adhesives due to their complications on migration testing and cost considerations. PP is used because it has good barrier properties, and high temperature resistance. Additionally, it is one of the lowest cost packaging films.
Storage Containers
In 1954, Guilio Natta developed Polypropylene (PP). Its versatility, in terms of both application and processing, has made it a great success. Its importance is in allowing a polymer to be made with specific tacticity.10 The Zieglar-Natta polymerization of propylene is shown in figure 8.

Figure 8: PP Polymerization10
Polypropylene
Polypropylene is a vinyl polymer from the polyolefin family, and was first produced in large scale in 1957.11 The ability of it to function as a fiber and as a thermoplastic has allowed it to be used to manufacturer many products in the durable and non-durable goods markets. The chemical structure of polypropylene is the result of the polymerization of propylene. The chemical chain structure of the two common forms of polypropylene can be seen in figure 9.
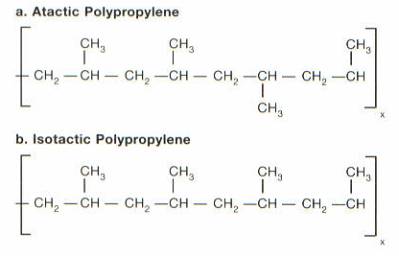
Figure 9: PP Chain Structure3
Atactic PP is characterized by its amorphous and tacky behavior, and lower molecular weight. It provides the same effects as a plasticizer, by reducing the crystallinity of the PP.3 Small amounts of atactic polymer in the final product results in improvements of certain mechanical properties, such as improved low temperature performance, elongation, and processability.
Isotactic PP is formed using a zinc catalyst, which exhibits high yield and high sterospecificity.11 Isotactic PP is the most stereo-regular structures of PP. The high sterospecificity results in a high degree of crystallinity. The increased crystallinity of PP makes it a good replacement for engineered thermoplastics (ETPs). The basic difference between PP and many engineered thermoplastics is that PP is a crystalline polymer where most ETPs are classified as amorphous polymers. Figure 10 shows the mechanical properties of the crystalline polymers versus amorphous polymers.
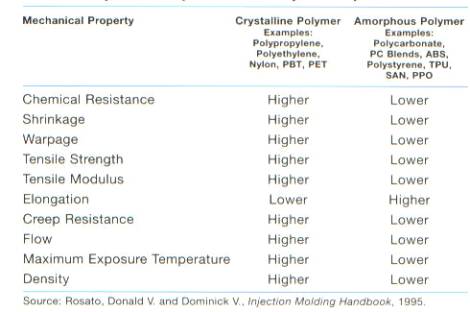
Figure 10: Mechanical Properties of Common Polymers13
Polypropylene, like many other thermoplastics, has its own unique characteristics. PP has a density ranging from .880 to .920 gram per cubic inch, which is lower than other engineered thermoplastics, therefore there is a potential for weight reduction.11 INSPIRE® PP produced by Dow Plastics has good heat resistance, and can be used in an constant environment of up to 2200F. INSPIRE® polypropylene is resistant to chemical attack from solvents, and chemicals in harsh environments. The common properties of polypropylene are presented in figure 11.
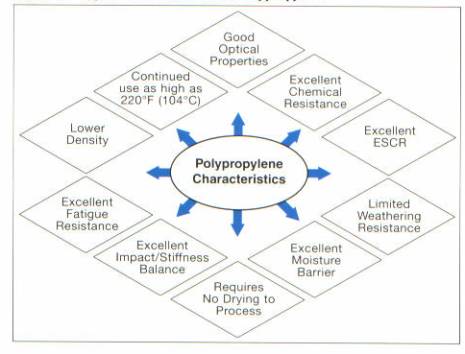
Figure 11: Typical Characteristics of
Polypropylene3
Processing
Manufacturing of polypropylene is performed in a variety of methods. However, food storage containers are normally manufactured by injection or blow molding. An injection-molded profile for polypropylene is illustrated in figure 12.3 In 2000, 1550 million pounds of PP packaging were produced at an approximate cost of .53 $/lb.5,11
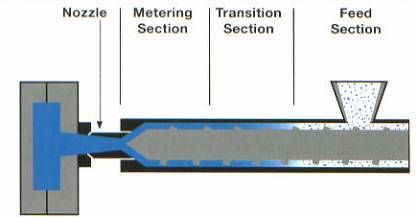
Figure 12: Injection molding barrel profile12
Regulations and Health Issues
The code of federal regulations provides exact guidance on the polymers allowed in the direct contact of food. Article 177 presents data specific to each polymer. This data includes: composition specifications, allowable coatings, emulsifiers, modifiers, and testing criteria. The following regulations are general rules for each polymer. Some of the information may not be pertinent to the actual products commercially used.
CPET
Article 177.1630 states the regulations for PET, produced by
condensation of either dimethyl terephthalate and ethylene glycol, or
terephthalic acid and ethylene glycol.7
The degree of crystallinity of PET is inconsequential.
The emulsifiers allowed in food suitable PET are sodium dodecylbenzenesulfonate,
sodium lauryl sulfate, and 2-sulfoethyl methacrylate. These compounds are used as an adjuvant in the application of
copolymer coatings.7 The
amount of the emulsifier should not exceed 2.0% of the dry weight of the
coating.7
PET specifications for oven cooking temperatures above 250°F are very straightforward. The PET surface, when exposed to distilled
water at 250°F for two hours
must yield less than 0.02 mg/in2 of chloroform-soluble extractives.7 In addition, when exposed to n-heptane at
150°F for two
hours, the chloroform-soluble extractives must not exceed 0.02 mg/in2
of food contact surface exposed.7
PPO/HIPS
PPO/HIPS is blended polymer where HIPS is the majority polymer. Article 177.1810 is the governing regulation for polystyrene graft polymers. This article regulates polystyrene graft polymers with 1,3-butadiene produced by catalytic polymerization of styrene and 1,3-butadiene.7 The minimum molecular weight is 29,000 and it must be completely soluble in toluene. In addition, the maximum extractable fractions of chloroform in distilled water and 50% ethanol are 0.0039 mg/cm2 of polymer surface for 0.19 cm thick sample.7 The distilled water extraction is performed at the reflux temperature for thirty minutes, while the ethanol extraction is carried out at 66 °C for two hours.
Even though HIPS is the majority component, the regulations only state requirements for HIPS that is a component the PPO. The regulations for PPO are located in article 177.2460. The intrinsic viscosity must not be less than 0.30 deciliter per gram of polymer. The extraction limit is 0.02 weight percent when extracted with n-heptane at 160°F for two hours.7 The extraction is performed with 200 milliliters of solvent and 25 grams of polymer.
Polypropylene
Article 177.1520 gives the regulations for olefin polymers, which
includes polypropylene. Polypropylene
food containers are made form catalytic polymerization of propylene with a
metallocene catylyst.7 Two
modifiers are allowed per federal regulations.
5-Ethylidine-2-norborene and 5-methylene-2-norborene may be used if not
to exceed 5% weight of the total polymer.7 There are thousands of possible additives to polypropylene. Additional adjuvant substances, not listed
in other regulations, possibly needed for processing the polypropylene are
located in article 177.1520, a few examples are petroleum hydrocarbon resins,
polyoxyethylene-grafted polydimethylsiloxane, and trisopropanolamine. Each additive has limitations also listed in
the regulations.
Specifications for propylene include a density of 0.880 to 0.913 and a
melting point of 160°C - 180°C.7 In addition, the
maximum extractable fraction of n-hexane is 6.4% at the reflux temperature, and
the maximum soluble fraction of xylene is 9.8% at 25°C.
For the n-hexane extraction, a sample of polypropylene is extracted at
50°C for two hours
and filtered.7 The filtrate
is then evaporated, and the residue is then weighed. The equation for percent n-hexane extractable is
![]()
The xylene experiment is different from that of n-hexane. A sample of polypropylene is dissolved in
100 milliliters of solvent at 120°C, then the sample is cooled and placed in a
constant water bath.7 40-50
milliliters are filtered through a vacuum filter, and a suitable volume of
aliquot from the filtrate is pipetted to the dish.7 The solvent is then evaporated and the
residue is weighed. The equation for
soluble xylene is
![]()
![]()
Testing
and Health Issues
These regulations are for polymers used as for food contact
surfaces. There are no specific
requirements for microwave food containers.
However, there is guidance on plastic containers in the microwave. The guidance is in the form of the FDA's
recommendations on chemistry information required for food contact
surfaces. Before a product goes to the
market, the FDA recommends that this guidance be used by a company to determine
proper migration testing protocol for their food container.
The guidance breaks down three different microwave container
applications. They are dual-oven trays,
microwavable containers, and microwave heat-susceptor packaging. Most PET microwavable containers serve as
dual-oven trays. Migration testing is
taken at the highest intended oven cooking temperature, at the longest intended
cooking time.12 The test
should use cooking oil as a food simulating liquid.12 These migration tests are done using food
simulants, since real food is difficult to analyze. Depending on the nature of the food to be packaged (i.e. acidic,
fatty), a different testing fluid and criteria must be used. The federal regulations previously discussed
talked of migration tests using a specific solvent, which are done in addition
to these tests.
Polypropylene containers are used only in the microwave. The FDA recommends that a company consult
with them before deciding a migration testing protocol for microwave only
containers.12 This is due to
the relative uncertainty involving the maximum temperature the container
reaches while in the microwave. Also,
size and type of food, cooking time, and food geometry effect container
temperature.
Migration is tested due to the effects that certain polymer additives
have on the human body. Some additives
are carcinogens, while plasticizers have been linked to endocrine
disorders. Further study is needed to
determine possible health risks due to migration of polymer additives.
Conclusions
The primary materials for microwavable food containers are polypropylene, crystallized polyethylene terephthalate, and polyphenylene oxide, high impact polystyrene blend. These selected polymers provide good temperature resistance, impact strength, lightweight, and minimal migration. For this reason, these polymers can cover consumer food applications ranging from single use home meal replacement to repeated use food storage containers.
Although there is a possibility of health risks, due to microwavable plastic food contact surfaces, modern testing and proper product design have essentially eliminated consumer hazards. Most problems occur when consumers use containers not suitable for microwave ovens, such as polystyrene.
The polymer industry continues to produce cost effective containers to keep up with the increasing consumer demand for quick prepared meals. The ability of a lightweight polymer to support loads greater than its weight demonstrates how valuable polymers are for cooking containers. Additionally, polymer packaging is significantly lighter than most alternatives, thus saving the food industry millions of dollars in transportation costs.
References
1.
Rubbright,
H.A., Davis N.O., The Microwave Decade, Packaging Strategies, West Chester, PA,
1989.
2.
Becker, R. “As
a Matter of Fact.” Chem Matters
April 2000: Volume 18 Number 2.
Online. Available HTTP: www.acs.org/education/curriculum/chemmatt.html
3. Dow Plastics, Polypropylene Resins Molding Guide. The Dow Chemical Company, 1998.
4.
Brady, A.L.,
Marsh, K.S., The Wiley Encyclopedia of Packaging Technology. New York: John Wiley and Sons, Inc., 1997.
5.
Hanlon, J.F.,
Kelsey, R.J., Forcinio, H.E., Handbook of Package Engineering. Technomic
Publishing Company, Inc., 1998.
6.
Selke, S.E., Understanding
Plastics Packaging Technology.
Cincinnati: Hanser/Gardner Publications, Inc., 1997.
7.
1999 CFR Title
21 Food and Drugs, Volume 3. Part 177
Indirect Food Additives: Polymers.
Online. Available HTTP: www.access.gpo.gov/nara/cfr/waisidx_99/21cfr177_99.html
8.
Eastman
Chemical Company. Online. Available HTTP: www.eastman.com/productfiles/prod0018.htm
9. General Electric Company. Plastics Division. Online. Available HTTP: www.geplastics.com/resins/techsolution/technifacts.html
10. Department of Polymer Science, University of Southern Mississippi, Macrogalleria, 1996. Online. Available HTTP: www.psrc.usm.edu/macrog/floor2.htm
11.
Modern Plastics Encyclopedia. New York: McGraw-Hill Companies, Inc., 2001.
12. U.S.
Food and Drug Administration. Online.
Available HTTP: vm.cfsan.fda.gov/~dms/opa-pmnc.html
13. U.S.
Food and Drug Administration. FDA
Consumer Magazine, Volume 97, Number 11.
Maryland; FDA, 1998.